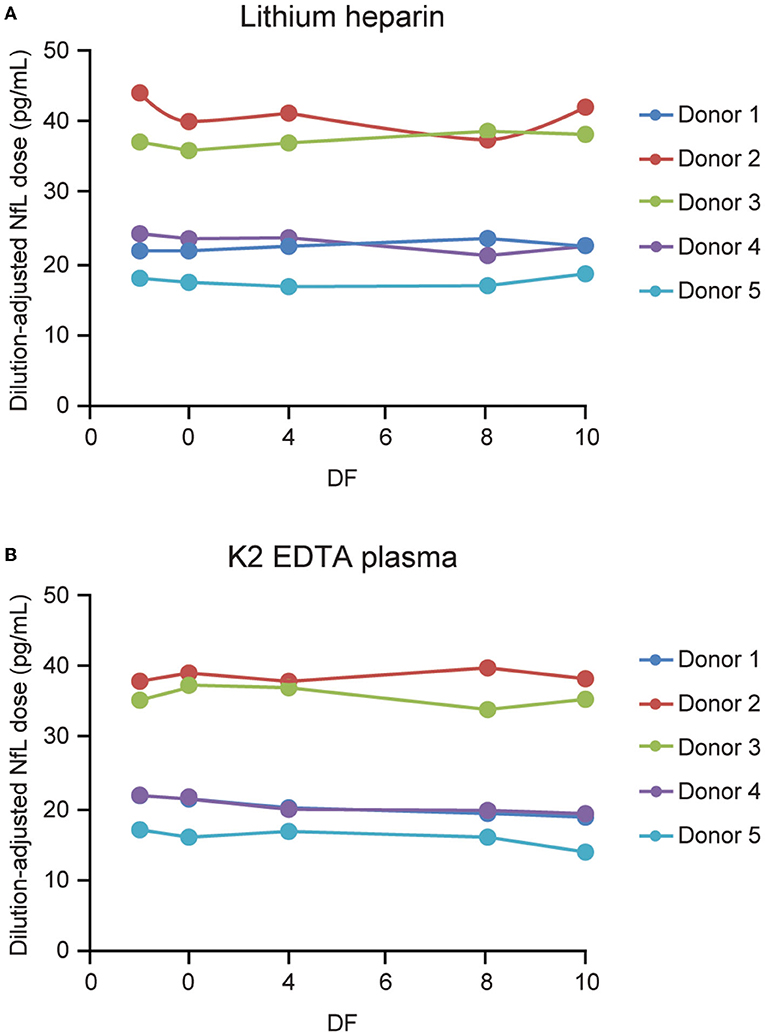
Frontiers | Development of a Highly Sensitive Neurofilament Light Chain Assay on an Automated Immunoassay Platform
Při střelbě v centru Paříže zemřeli tři lidé, další jsou zranění. Podezřelý útočil už v minulosti | iROZHLAS - spolehlivé zprávy

Těžký válečný zločin.“ Nemocnice zasažená americkou raketou? Moskva obvinila Kyjev | ParlamentniListy.cz – politika ze všech stran

Effects of a fixed combination of perindopril and indapamide on macrovascular and microvascular outcomes in patients with type 2 diabetes mellitus (the ADVANCE trial): a randomised controlled trial - The Lancet

Subcutaneous versus intravenous administration of (neo)adjuvant trastuzumab in patients with HER2-positive, clinical stage I–III breast cancer (HannaH study): a phase 3, open-label, multicentre, randomised trial - The Lancet Oncology

Moskva se snaží zničit ukrajinské elektrárny. Rakety zasáhly asi 30 % energetické infrastruktury - Echo24.cz

U Moskvy roste luxusní klinika pro kremelské prominenty. Ti kvůli sankcím nemohou na léčbu do ciziny - Aktuálně.cz














/cloudfront-us-east-2.images.arcpublishing.com/reuters/JNZBUZK3DBOYJI7DMVU4LKTACM.jpg)


